Research Article
Vitamin D Predicts Synopsis of Fatigue and Quality of Life in Advanced Cancer
- Hany N. Azzam
Corresponding author: Hany N. Azzam, Department of Pharmacology and Toxicology, Heliopolis University, Faculty of Pharmacy, Cairo, Egypt.
Volume: 3
Issue: 1
Article Information
Article Type : Research Article
Citation : Hany N. Azzam, Vitamin D Predicts Synopsis of Fatigue and Quality of Life in Advanced Cancer. Journal of Medicine Care and Health Review 3(1). https://doi.org/10.61615/JMCHR/2026/APRIL027140404
Copyright: © 2026 Hany N. Azzam. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.61615/JMCHR/2026/APRIL027140404
Publication History
Received Date
10 Mar ,2026
Accepted Date
21 Mar ,2026
Published Date
04 Apr ,2026
Abstract
Background
The purpose of this study is to evaluate the association between parathyroid hormone (PTH) and vitamin D insufficiency and health-related QOL problems, weariness, and physical functioning in Egyptian patients with advanced cancer.
Methods
Enzyme-linked immunosorbent assay (ELISA) was used to quantify serum vitamin D and PTH levels in 58 Egyptian patients with advanced cancer compared to 22 control persons. The Functional Assessment of Chronic Illness Therapy (FACIT) version 4 palliative care questionnaire's tiredness subscale was used to measure cancer-related fatigue, while four primary areas (fatigue, QOL, physical dysfunction, and appetite loss) were measured using the European Organization for Research and Treatment of Cancer Quality of Life (EORTC QLQ-C15-PAL) version 1 questionnaire. Additionally, the Palliative Performance Scale (PPS) was used to calculate performance ratings. Anthropometric characteristics were assessed.
Results
The study was conducted on cancer and control groups defined as follows:
Group 1 had 58 advanced cancer patients.
Group (2): 22 healthy participants were recruited from the community through a notice.
Group (1) had lower serum vitamin D levels [5.74(5.07), p˂0.0001] compared to group (2) [28.1(8.1), p˂0.0001], but group (1) had higher serum intact PTH levels [82(21.7), p˂0.0001] compared to group (2) [33.9(29.2), p˂0.0001].
Conclusion
Egyptian patients with advanced cancer frequently had low vitamin D levels, which were linked to severe tiredness symptoms, poor performance, and a terrible quality of life. There was an inverse relationship between serum intact PTH levels and serum vitamin D levels. Egyptian patients with advanced cancer frequently had high PTH levels, which were associated with severe tiredness symptoms, poor performance, and a negative quality of life.
Key words: Vitamin D, Parathyroid hormone (PTH), Quality of life (QOL), Fatigue.
►Vitamin D Predicts Synopsis of Fatigue and Quality of Life in Advanced Cancer
Hany N. Azzam1*
1Department of Pharmacology and Toxicology, Heliopolis University, Faculty of Pharmacy, Cairo, Egypt.
Background
A vital hormone with a variety of roles, vitamin D is necessary for good health. Diseases involving immunological, cardiovascular, and respiratory system dysregulation have been linked to vitamin D. The most common method for determining if vitamin D reserves are adequate is to measure levels of 25-Hydroxy Vitamin D (25(OH) D). It is uncertain what the size of the impact of vitamin D deficiency is on severely unwell individuals [1].
Most people get the vitamin D their bodies need via sporadic exposure to sunshine. The skin produces enough cholecalciferol (vitamin D3) in the spring, summer, and fall to store it in body fat. This fat is subsequently stored and organized throughout the winter, when the skin produces little to no vitamin D3 [2]. The foods provide a minimal quantity of vitamin D. There are only a few foods that naturally contain considerable levels of vitamin D. These include sun-exposed mushrooms, fatty fish like tuna, salmon, and sardines, and the oils from some fish's livers, such as cod [3].
Vitamin D insufficiency was linked to low milk consumption, central obesity, and non-use of vitamin D supplements, indicating potentially significant factors for preventative intervention [4]. Vitamin D is quite low across age groups in the Middle East region.
Older age, feminine gender, multiparity, conservative attire, poor socioeconomic class, and urban life are all reliable indicators of low levels [5]. Prostate cancer was the most prevalent illness among males worldwide in 2018, while breast cancer was the most common cancer incidence among women [6]. In less developed nations, cancer presently accounts for 57% of cases and 65% of cancer-related deaths globally [7].
The most common symptom of cancer is fatigue, which is associated with substantial morbidity, functional impairments, and a worse quality of life (QOL). As a result, managing tiredness effectively will significantly lower the illness burden related to cancer and its therapies [8].
From the beginning of carcinogenesis to metastasis and interactions between cells and the milieu, vitamin D can control every stage of the process [9].
A lack of vitamin D raises the chance of getting cancer and dying from it [10], New studies point to vitamin D's possible role in stopping the spread of cancer and lowering its side effects, including arthralgia, muscular weakness, and bisphosphonate toxicity [11-14] [15-17] [18]. Thus, a lack of vitamin D is linked to several symptoms, such as discomfort and exhaustion. These signs are typical of advanced cancer [19], where normalization of vitamin D levels was shown to improve fatigue in advanced cancer patients [20].
Reduced intestinal calcium (Ca2+) absorption, increased parathyroid hormone (PTH) release, and accelerated bone resorption are all caused by low 25 (OH) D [21]. PTH is widely employed as a biomarker of classical vitamin D physiology in human clinical studies. According to certain cross-sectional studies, there is a stronger correlation between PTH and bioavailable or free 25 (OH) D [22, 23]. Increased PTH preserves a normal blood Ca2+ content in vitamin D insufficiency at the skeleton's price [24]. Fatigue, discomfort, memory and concentration issues, irritability, melancholy, anxiety, and sleep issues are among the symptoms reported by patients with primary hyperparathyroidism (HPT) [25].
The primary aim of this study was to assess the relationship between Vitamin D deficiency and health-related quality-of-life issues, fatigue, and physical functioning in advanced cancer Egyptian patients.
The secondary aim of this study was divided into two parts. The first part includes assessing the relationship between Vitamin D and PTH. In contrast, the second section examined the association between PTH and physical functioning, weariness, and health-related quality-of-life difficulties in Egyptian patients with advanced cancer.
Subjects and Methods
Subject Recruitment and Study Design
There were 80 male and female subjects enrolled in this study; 22 of them were healthy volunteers without cancer, and they served as control subjects.
The remaining 58 were adults who have locally advanced or metastatic cancer with an assessed life-expectancy of at least 4 weeks, a normal body mass index (BMI) level, and PPS > 30%.
Serum samples from adults who had been enrolled in this study were collected from oncology outpatient clinics, Al Demerdash hospital, and Ain Shams University (ASU) under physician supervision.
Exclusion criteria for advanced cancer Egyptian patients and normal control subjects included the following: a) History of Ca2+ or vitamin D supplements the last three months; b) Chronic renal and/or liver disease; c) Patients taking anticonvulsants, glucocorticoids or immunosuppressant drugs; d) Having received chemotherapy or radiation therapy within the last 3 weeks prior to inclusion to avoid their impact on fatigue; e) PPS < or = 30%; f) Patients with BMI < 18.5 or > 30.
After the collection of blood samples and centrifugation, yielded serum was divided into 2 portions and kept in aliquots at -80ºC for subsequent use.
Quantification of 25-Hydroxy Vitamin D Concentration in Advanced Cancer Egyptian Patients’ Serum using 25-Hydroxy Vitamin D Enzyme-Linked Immunosorbent Assay Kit
In the first portion, the blood vitamin D concentration was measured using the ELISA technique in compliance with the manufacturer's instructions using a commercially available kit from Bioassay Technology Laboratory (China) (Cat # E1981Hu) [26]. The absorbance unit optical density (OD) at 450 nanometers (nm), recorded in a microplate reader, was plotted against concentration using the values derived from the standard to create a linear regression dose-response curve. This curve was used to calculate the samples' 25 (OH) D concentration.
Quantification of Intact Parathyroid Hormone Concentration in Advanced Cancer Egyptian Patients Using Parathyroid Hormone Enzyme-Linked Immunosorbent Assay Kit
In the second section, a commercially available kit provided by IBL International GMBH (Germany) was used to measure the concentration of serum intact PTH using the ELISA technique in accordance with the manufacturer's instructions (Cat # NM59041) [27]. A 4-parameter logistical curve was created by graphing the following data: absorbance unit OD at 450 nm, measured in a microplate reader, vs. concentration, calculated using the standard values. The concentration of PTH in the samples was measured using this curve.
Assessment of Symptoms of Quality of Life In Advanced Cancer Egyptian Patients Through the European Organization for Research and Treatment of Cancer Quality of Life Palliative Care Questionnaire (EORTC QLQ-C15-PAL) Version 1
It is a simplified 15-item version of the EORTC QLQ-C30 (version 3.0) designed for palliative care. Two strategies were employed to produce the EORTC QLQ-C15-PAL. The first technique included reducing multi-item scales using item response theory and building algorithms to score the smaller scales using the original response scale metric. The second strategy comprised interviewing patients and health care professionals to identify scales and/or individual items that were unsuitable or not highly relevant and hence may be removed. Algorithms are used to estimate the QLQ-C15-PAL scores for four shortened measures that measure physical functioning, emotional functioning, nausea and vomiting, and exhaustion [28]. In this study, we calculated raw scores for four domains of the questionnaire as follows (Raw score = total score of questions in each domain / no. of questions): Quality of life domain, Fatigue domain, Physical functioning domain, and Appetite loss domain. The questionnaire and its development, which was described in several references [29-32], is shown clearly in Table 1 [33].
Table 1: EORTC QLQ-C15-PAL (Version 1) Questionnaire
|
We are interested in some things about you and your health. Please answer all of the questions yourself by circling the number that best applies to you. There are no "right" or "wrong" answers. The information that you provide will remain strictly confidential |
||||
|
Please fill in your initials: |
||||
|
Your birthdate (Day, Month, Year): Today's date (Day, Month, Year): |
||||
|
Question |
Not at all (1) |
A little (2) |
Quite a bit (3) |
Very much (4) |
|
1. Do you have any trouble taking a short walk outside of the house? |
1 |
2 |
3 |
4 |
|
2. Do you need to stay in bed or a chair during the day? |
1 |
2 |
3 |
4 |
|
3. Do you need help with eating, dressing, washing yourself, or using the toilet? |
1 |
2 |
3 |
4 |
|
During the past week: |
Not at all (1) |
A little (2) |
Quite a bit (3) |
Very much (4) |
|
4. Were you short of breath? |
1 |
2 |
3 |
4 |
|
5. Have you had pain? |
1 |
2 |
3 |
4 |
|
6. Have you had trouble sleeping? |
1 |
2 |
3 |
4 |
|
7. Have you felt weak? |
1 |
2 |
3 |
4 |
|
8. Have you lacked appetite? |
1 |
2 |
3 |
4 |
|
9. Have you felt nauseated? |
1 |
2 |
3 |
4 |
|
10. Have you been constipated? |
1 |
2 |
3 |
4 |
|
11. Were you tired? |
1 |
2 |
3 |
4 |
|
12. Did pain interfere with your daily activities? |
1 |
2 |
3 |
4 |
|
13. Did you feel tense? |
1 |
2 |
3 |
4 |
|
14. Did you feel depressed? |
1 |
2 |
3 |
4 |
|
For the following question, please circle the number between 1 and 7 that best applies to you |
||||
|
15. How would you rate your overall quality of life during the past week? |
1 – 7 ( ) 1: Poor 7: Excellent |
|||
Assessment of Cancer-Related Fatigue in Advanced Cancer Egyptian Patients Through the Fatigue Subscale of the Functional Assessment of Chronic Illness Therapy (FACIT) Version 4 Palliative Care Questionnaire
This is a widely used 13-item fatigue sub-scale where each item is a 5-point Likert self-reported scale ranging from 0=“not at all” to 4=“very much so.” The total score varies from 0=“worst condition” to 52 = “best condition” [34]. This is clearly shown in Table 2 [35]. All FACIT scales are assessed, and a high score indicates strong performance. To do this, response scores to negatively worded questions were inverted, and item responses were averaged. When individual questions are skipped, scores are judged using the average of the other responses on the scale [36].
Table 2: The FACIT (Version 4) Questionnaire
|
Below is a list of statements that other people with your illness have said are important. Please circle or mark one number per line to indicate your response as it applies to the past 7 days |
|||||
|
Question |
Not at all (0) |
A little bit (1) |
Somewhat (2) |
Quite a bit (3) |
Very much (4) |
|
I feel fatigued |
0 |
1 |
2 |
3 |
4 |
|
I feel weak all over |
0 |
1 |
2 |
3 |
4 |
|
I feel listless (“washed out”) |
0 |
1 |
2 |
3 |
4 |
|
I feel tired |
0 |
1 |
2 |
3 |
4 |
|
I have trouble starting things because I am tired |
0 |
1 |
2 |
3 |
4 |
|
I have trouble finishing things because I am tired |
0 |
1 |
2 |
3 |
4 |
|
I have energy |
0 |
1 |
2 |
3 |
4 |
|
I am able to do my usual activities |
0 |
1 |
2 |
3 |
4 |
|
I need to sleep during the day |
0 |
1 |
2 |
3 |
4 |
|
I am too tired to eat |
0 |
1 |
2 |
3 |
4 |
|
I need help doing my usual activities |
0 |
1 |
2 |
3 |
4 |
|
I am frustrated by being too tired to do the things I want to do |
0 |
1 |
2 |
3 |
4 |
|
I have to limit my social activity because I am tired |
0 |
1 |
2 |
3 |
4 |
Calculation of Performance Scores for Advanced Cancer Egyptian Patients Through the Palliative Scale (PPS Performance) Version 2
After assessing the patients' QOL using the EORTC QLQ-C15-PAL (version 1) and the FACIT (version 4) questionnaires, PPS (version 2) was used to find a best-fit performance score for every subject. Several articles on the reliability and validity of the PPS have been published [37-42].
Statistical Analysis
GraphPad prism® version 6.02 and Microsoft Excel 2016 were used for data analysis and chart development. Values were reported as mean ± SEM, median, interquartile range (IQR), and percentage. To assess the normal distribution pattern, the D'Agostino and Pearson omnibus normality tests were used on the data from the groups and subgroups. However, the data for serum vitamin D and PTH levels did not pass the normality test after subgrouping PPS percentages; the Kruskal-Wallis test was applied. Furthermore, the Mann-Whitney (U) tests were employed to identify significant differences in vitamin D and serum intact PTH levels in patients and healthy controls across groups and subgroups, where appropriate. Spearman's rho (r) was used to calculate the correlation between two variables.
Results
Serum Vitamin D Levels
It was found that serum vitamin D levels were deficient in cancer patients [5.74(5.07), p˂0.0001] with statistically significant difference in comparison to age-matched control subjects [28.1(8.1), p˂0.0001] [31.9(43.9), as shown in Figure (1) and Table (3).
Figure 1: The Boxplot (Median and IQR) for the Serum Vitamin D Levels Among Cancer Patients and the Control Group
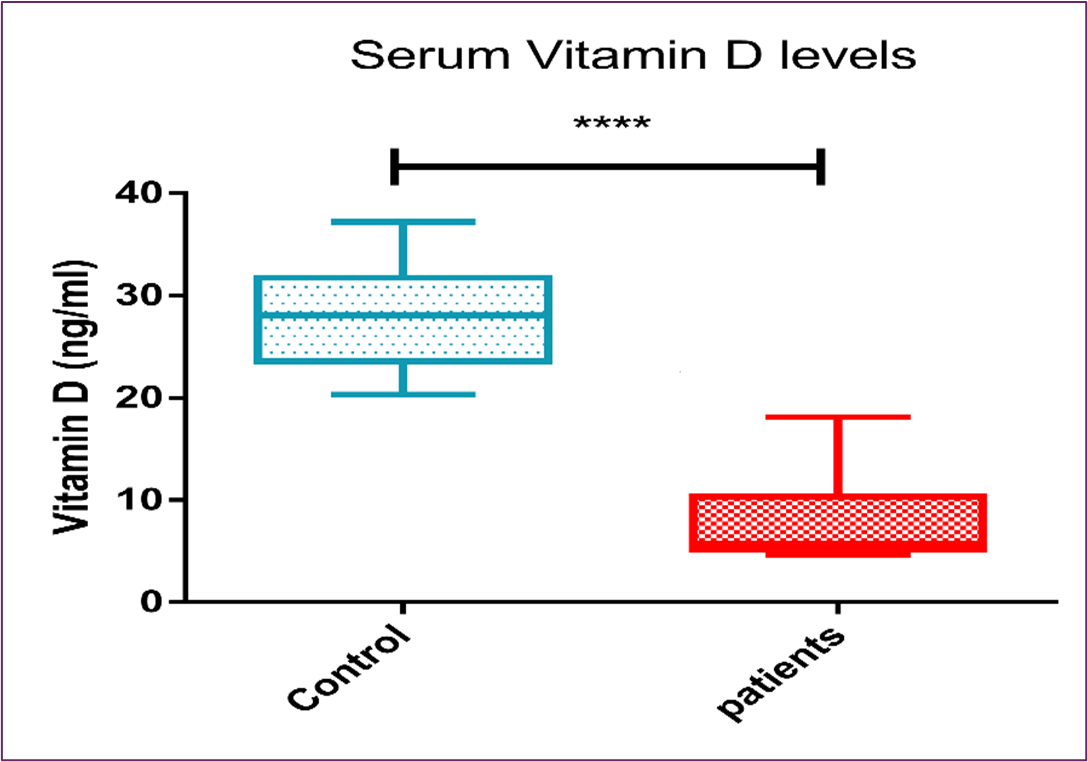
Table 3: Serum Vitamin D levels
|
Serum Vitamin D |
N |
Range |
Mean |
SD |
Median (IQR) |
P |
|
|
Min |
Max |
||||||
|
Cancer patients |
58 |
4.62 |
18.1 |
8.27 |
4.10 |
5.74 (5.07) |
<0.0001**** |
|
Control group |
22 |
20.3 |
37.2 |
27.9 |
5.05 |
28.1 (8.1) |
<0.0001**** |
Serum Intact Parathyroid Hormone Levels
It was found that serum intact PTH levels were elevated in cancer patients [82(21.7), p˂0.0001] with statistically significant difference in comparison to age-matched control subjects [33.9(29.2), p˂0.0001], as shown in Figure (2) and Table (4).
Figure 2: The Boxplot (Median and IQR) for the Serum Intact PTH Levels Among Cancer Patients and the Control Group
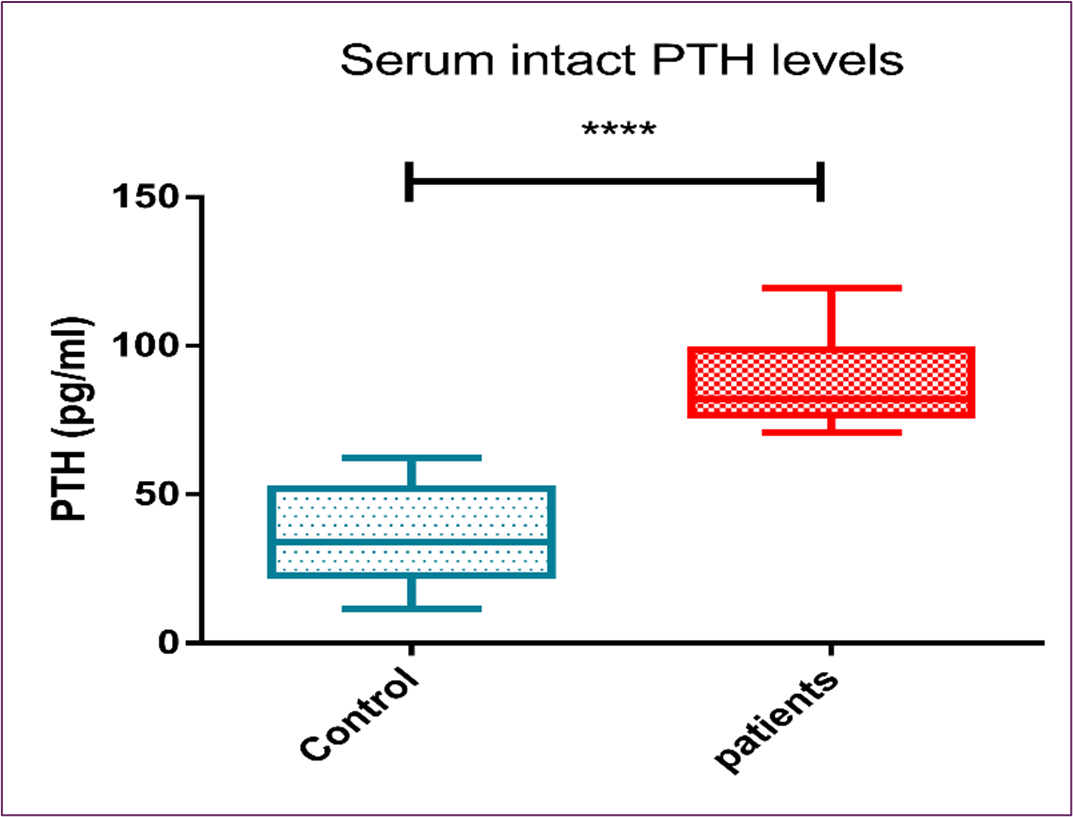
Table 4: Serum Intact PTH levels
|
Serum PTH |
N |
Range |
Mean |
SD |
Median (IQR) |
P |
|
|
Min |
Max |
||||||
|
Cancer patients |
58 |
70.7 |
120 |
87.6 |
13.5 |
82 (21.7) |
<0.0001**** |
|
Control group |
22 |
11.4 |
62.4 |
36.2 |
16.1 |
33.9 (29.2) |
<0.0001**** |
Correlations
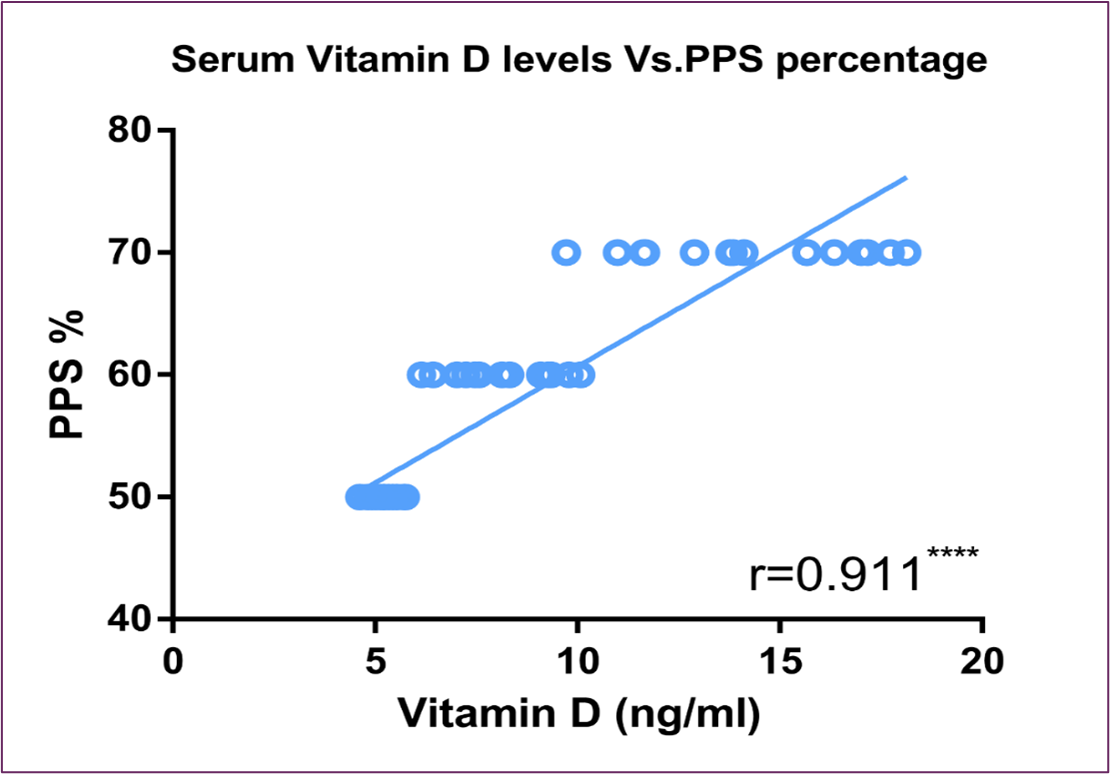
Serum vitamin D levels exhibited a substantial inverse correlation with serum intact PTH, as shown in Figure (3). On the other hand, it showed significant direct correlation with FACIT scores and PPS percentage, as shown in Figure (4) and Figure (5) respectively.
Figure 3: Correlation Between Serum Vitamin D Level and Serum Intact PTH Level
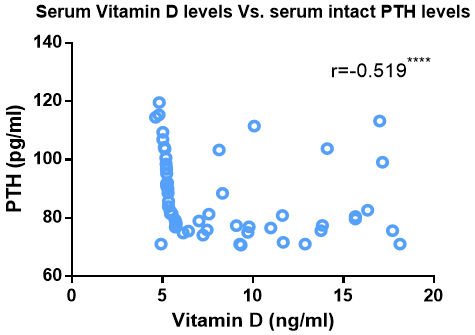
Figure 4: Correlation Between Serum Vitamin D Level and FACIT Score
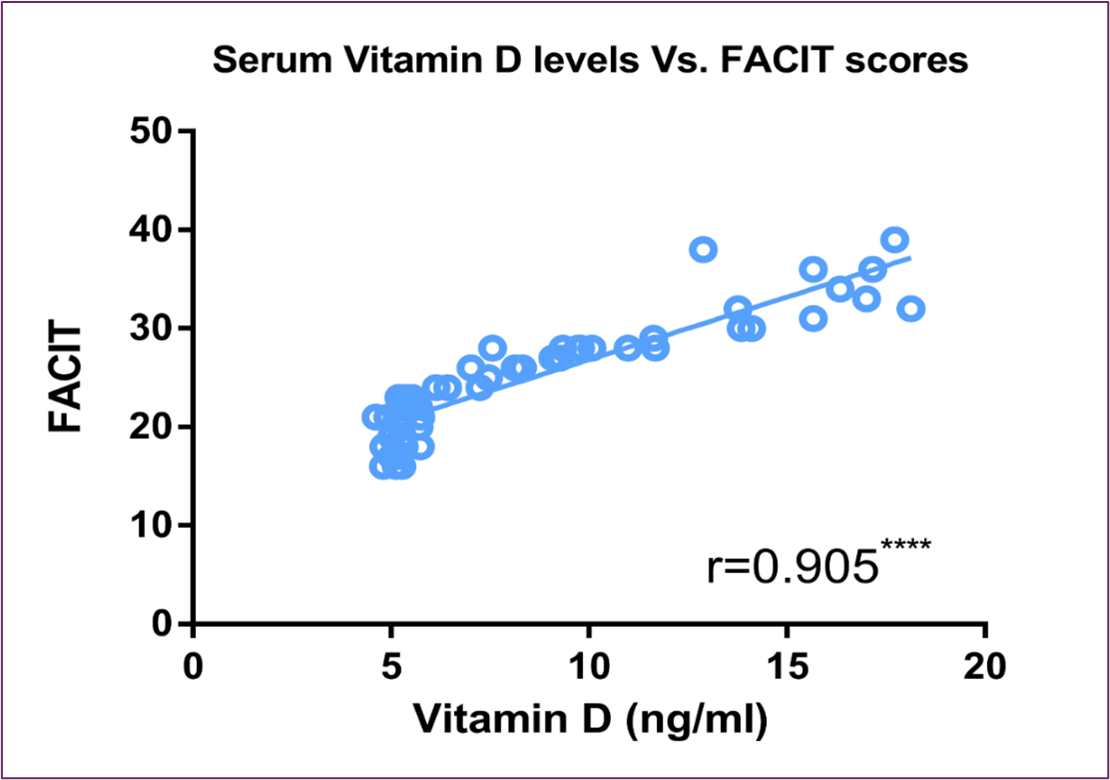
Figure 5: Correlation Between Serum Vitamin D Level and PPS Percentage
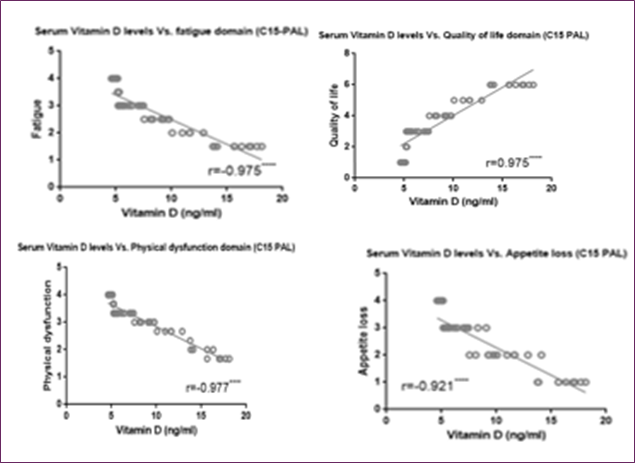
Moreover, Figure (6) showed a direct correlation between vitamin D and QOL C15 PAL score and an inverse correlation between vitamin D and fatigue, physical dysfunctioning and appetite loss C15 PAL scores.
Figure 6: Correlation between serum Vitamin D level and EORTC QLQ-C15-PAL different domains score (quality of life, fatigue, physical dysfunction, and appetite loss)
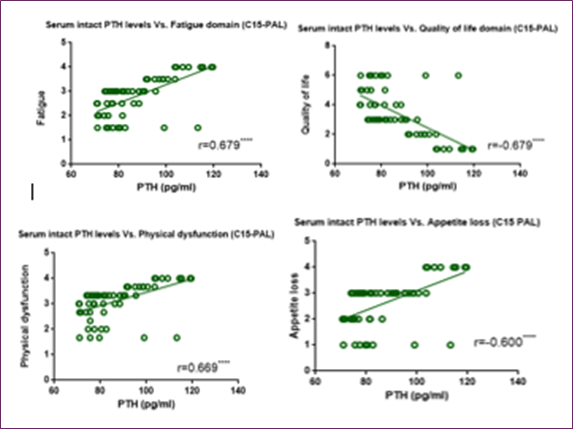
Concerning serum intact PTH level, Figure (7) and Figure (8) showed significant inverse correlation with FACIT scores and PPS percentage, respectively. Furthermore, Figure (9) showed an inverse correlation between PTH and QOL C15 PAL score and a direct correlation between PTH and fatigue, physical dysfunctioning and appetite loss C15 PAL scores.
Figure 7: Correlation Between Serum Intact PTH Level and FACIT Score
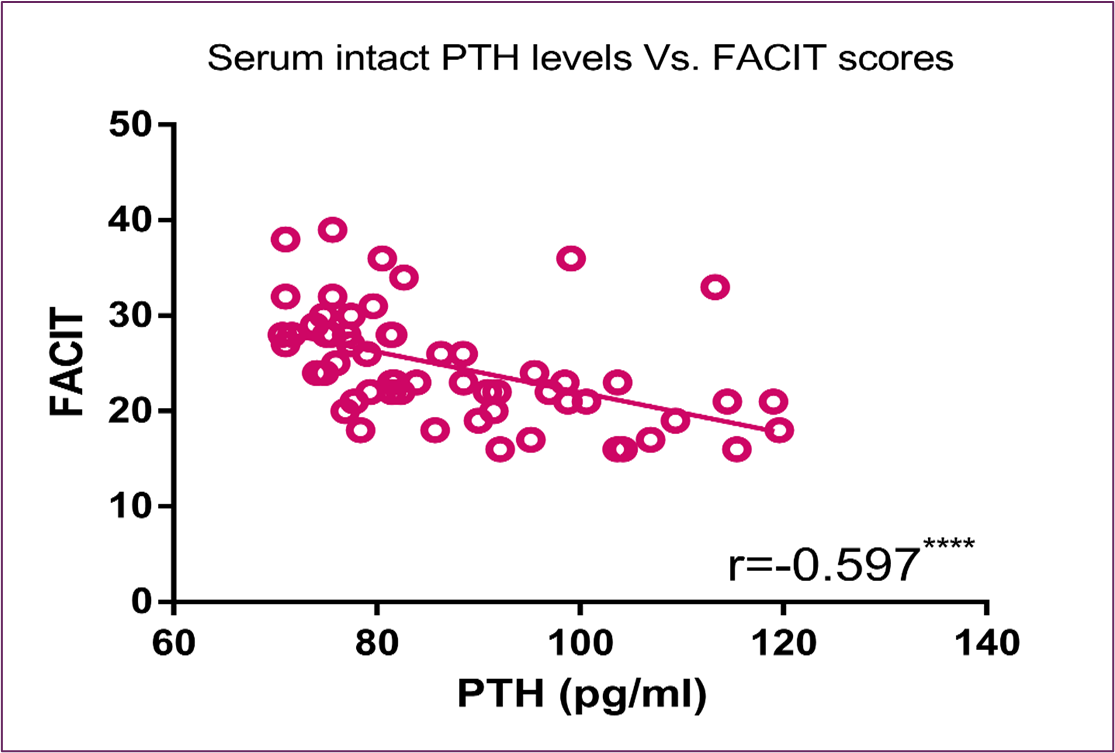
Figure 8: Correlation Between Serum Intact PTH Level and PPS Percentage
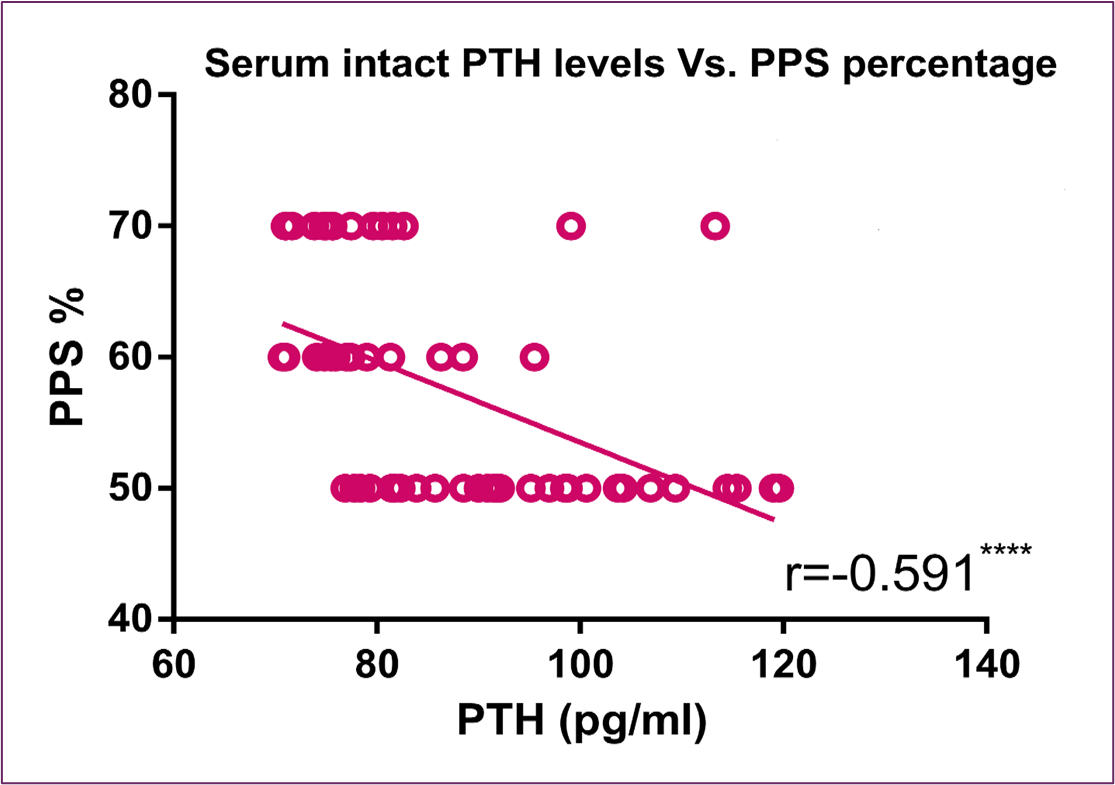
Figure 9: Correlation between serum PTH level and EORTC QLQ-C15-PAL different domains score (quality of life, fatigue, physical dysfunction, and appetite loss)
Discussion
Due to increased awareness and acceptance, vitamin D's function has changed from that of a vitamin to that of a crucial hormone. Vitamin D insufficiency persists in most regions of the world and across most age groups, even though many nations have sufficient solar exposure. Reduced synthesis, decreased absorption, and enhanced catabolism are the causes of this [43].
Vitamin D uses its nuclear receptor VDR to start pathways that control a number of bodily functions, including myocyte development and proliferation [44]. According to recent research, vitamin D insufficiency is linked to a number of chronic illnesses, including cancer and cardiovascular disease [45, 46].
Through anti-inflammatory and antioxidant defenses as well as DNA damage repair activities, vitamin D has been shown to have a significant role in avoiding the tumor starting stage [9, 47]. According to a number of epidemiological, clinical, preclinical, and in vitro experimental findings, vitamin D signaling activation may be a viable preventative and therapeutic approach for several cancer types [9]. Growth suppression occurs when prostate, colon, breast, lung, and melanoma cancer cell lines are exposed to vitamin D3 in vitro, indicating a potential therapeutic function for vitamin D in cancer treatment, as well as a link between vitamin D deficiency and cancer risk and development [48].
As far as we know, this is the first research in Egypt and the Middle East to employ FACIT, EORTC C-15 PAL, and PPS scores to determine a link between vitamin D and PTH and fatigue symptoms and quality of life in advanced cancer patients.
According to a recent case-control study on the impact of vitamin D on breast cancer in Ethiopian women, vitamin D insufficiency is more prevalent in Ethiopian women with breast cancer [49]. Another research on 88 males with histologically confirmed prostate cancer revealed that patients with prostate cancer had worse vitamin D status than control non-cancerous individuals, as seen by significantly lower levels of total 25 (OH) D and bioavailable 25 (OH) D [50]. Vitamin D insufficiency was prevalent among American cancer patients with old solid tumors, according to another retrospective cohort research [51].
These findings worked in alignment with our study, where Figure (1) revealed statistically significant vitamin D deficiency with levels < 20 ng/ml in advanced cancer Egyptian subjects compared to age-matched healthy controls.
Another finding in our study was that PTH showed statistically significant elevation in advanced cancer Egyptian patients compared to age-matched healthy control subjects. The study of cancer patients revealed a substantial negative association between a drop in vitamin D levels and an increase in PTH levels, as shown in Figure (2) and Figure (3) respectively. These results dovetail with the early published data that associated increased PTH with vitamin D deficiency, and hence maintaining a normal serum Ca2+ concentration at the expense of the skeleton [24].
According to a recent study, vitamin D supplementation was linked to greater levels of self-reported health-related QOL and may help women with breast cancer avoid recurrence, extend life, and enhance their mood [52]. Another study on women with a history of invasive breast cancer between the ages of 50 and 80 revealed that vitamin D usage after diagnosis was linked to a lower death rate from breast cancer [53]. Additionally, there is mounting evidence that vitamin D supplementation may enhance quality of life and lessen depressed symptoms [54, 55]. A recent study of published randomized clinical trials (RCTs) found that vitamin D supplementation significantly lowered the mean pain score in cancer patients with chronic pain when compared to placebo medication [56].
The idea that a large percentage of patients with advanced cancer in palliative care have vitamin D deficiency, or 25(OH)D<20ng/mL, and that there is a relationship between serum vitamin D levels and the patient's self-assessment of QOL as well as the patient's ability to perform daily activities, stems from the significant fatigue, physical, and functional disability in these patients, as well as the very few therapeutic measures readily available to reduce them [30]. For example, individuals with breast cancer who had greater blood 25(OH) D concentrations also had reduced death rates; patients with the greatest concentration of 25(OH) D had around half the death rate of those with the lowest concentration [57]. Similar associations were found in our study, where vitamin D showed positive correlation with fatigue score, QOL-C15 PAL domain score, and performance level percentage, and showed negative correlation with fatigue, physical dysfunction, and appetite loss C15 PAL domain scores as shown in Figure (4), Figure (5) and Figure (6) respectively.
On the contrary, PTH showed a negative correlation with fatigue score, QOL-C15 PAL domain score, and performance level percentage, and showed a positive correlation with fatigue, physical dysfunction, and appetite loss C15 PAL domain scores as shown in Figure (7), Figure (8) and Figure (9), respectively.
Conclusion
Egyptian patients with advanced cancer frequently had low vitamin D levels, which were linked to severe tiredness symptoms, poor performance, and a terrible quality of life. There was an inverse relationship between serum intact PTH levels and serum vitamin D levels. Egyptian patients with advanced cancer frequently had high PTH levels, which were associated with severe tiredness symptoms, poor performance, and a negative quality of life. This indicates that vitamin D insufficiency plays a critical role in the decline of quality of life and the aggravation of tiredness in Egyptian patients with advanced cancer; normalization of vitamin D levels has been demonstrated to alleviate fatigue in these patients [20].
Funding: This manuscript was prepared without any funding.
Code Availability: Not applicable.
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethics Approval: This study was conducted in accordance with the Declaration of Helsinki and reviewed by the ethical committee of the Faculty of Medicine in Ain Shams University, Cairo, Egypt, under Federal Wide Assurance number (FWA 000017585). After being educated about the investigations, study procedure, possible dangers, goal, and advantages of the study, all research participants signed an informed consent form.
Consent to Participate: Not applicable.
Consent for Publication: Not applicable
Clinical Trial Number: Not applicable
Availability of Data and Materials
The data for this study could be available from the corresponding authors upon reasonable request
Funding
Not Applicable
Copyright © The Author(s) 2025.
Abbreviations
|
25 (OH) D |
25-hydroxy vitamin D |
|
ASU |
Ain Shams University |
|
BMI |
Body Mass Index |
|
Ca2+ |
Calcium |
|
ELISA |
Enzyme-Linked Immunosorbent Assay |
|
EORTC-QLQ-C15 PAL |
European Organization for Research and Treatment of Cancer Quality of Life palliative care Questionnaire |
|
FACIT |
Functional Assessment of Chronic Illness Therapy |
|
HPT |
Hyperparathyroidism |
|
IQR |
Interquartile range |
|
nM |
Nanomolar |
|
OD |
Optical density |
|
PPS |
Palliative Performance Scale |
|
PTH |
Parathyroid Hormone |
|
QOL |
Quality of Life |
|
RCTs |
Randomized clinical trials |
|
Vitamin D3 |
Cholecalciferol |
- de Haan, K. (2014). Vitamin D deficiency as a risk factor for infection, sepsis and mortality in the critically ill: systematic review and meta-analysis. Critical Care. 18(6): 660.
- Holick, M.F. (2004). Vitamin D: importance in the prevention of cancers, type 1 diabetes, heart disease, and osteoporosis. The American journal of clinical nutrition. 79(3): 362-371.
- Muscogiuri, G. (2018). Vitamin D: past, present and future perspectives in the prevention of chronic diseases. European journal of clinical nutrition. 72(9): 1221-1225.
- AlQuaiz, A.M. (2018). Age and gender differences in the prevalence and correlates of vitamin D deficiency. Archives of osteoporosis. 13(1): 49.
- Mithal, A. (2009). Global vitamin D status and determinants of hypovitaminosis D. Osteoporosis international. 20(11): 1807-1820.
- Bray, F. (2018). Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer journal for clinicians. 68(6): 394-424.
- Torre, L.A. (2015). Global cancer statistics, 2012. CA: a cancer journal for clinicians. 65(2): 87-108.
- Wagner, L. and D. Cella. (2004). Fatigue and cancer: causes, prevalence and treatment approaches. British journal of cancer. 91(5): 822-828.
- Giammanco, M. (2015). Vitamin D in cancer chemoprevention. Pharmaceutical biology. 53(10): 1399-1434.
- Giovannucci, E. (2006). Prospective study of predictors of vitamin D status and cancer incidence and mortality in men. Journal of the National Cancer Institute. 98(7): 451-459.
- Drake, M.T. (2010). Vitamin D insufficiency and prognosis in non-Hodgkin's lymphoma. Journal of clinical oncology. 28(27): 4191-4198.
- Shanafelt, T.D. (2011). Vitamin D insufficiency and prognosis in chronic lymphocytic leukemia. Blood. 117(5): 1492-1498.
- Goodwin, P.J. (2009). Prognostic effects of 25-hydroxyvitamin D levels in early breast cancer. Journal of Clinical Oncology. 27(23): 3757-3763.
- Ren, C. (2012). Prognostic effects of 25-hydroxyvitamin D levels in gastric cancer. Journal of translational medicine. 10(1): 16.
- Amir, E. (2010). A phase 2 trial exploring the effects of high‐dose (10,000 IU/day) vitamin D3 in breast cancer patients with bone metastases. Cancer. 116(2): 284-291.
- Prieto-Alhambra, D. (2011). Vitamin D threshold to prevent aromatase inhibitor-induced arthralgia: a prospective cohort study. Breast cancer research and treatment. 125(3): 869-878.
- Waltman, N.L. (2009). Vitamin D insufficiency and musculoskeletal symptoms in breast cancer survivors on aromatase inhibitor therapy. Cancer nursing. 32(2): 143-150.
- Vaughan-Shaw, P. (2017). The impact of vitamin D pathway genetic variation and circulating 25-hydroxyvitamin D on cancer outcome: systematic review and meta-analysis. British journal of cancer. 116(8): 1092.
- Dorman, S. (2018). 169 Vitamin d supplementation for adults with advanced cancer: impact on quality of life, pain and fatigue. British Medical Journal Publishing Group.
- Roy, S. (2014). Correction of low vitamin D improves fatigue: effect of correction of low vitamin D in fatigue study (EViDiF Study). North American journal of medical sciences. 6(8): 396-402.
- Holick, M.F. (2007). Vitamin D deficiency. New England Journal of Medicine. 357(3): 266-281.
- Bhan, I. (2012). Bioavailable vitamin D is more tightly linked to mineral metabolism than total vitamin D in incident hemodialysis patients. Kidney international. 82(1): 84-89.
- Schwartz, J.B, L. Kane, and D. Bikle. (2016). Response of Vitamin D Concentration to Vitamin D3 Administration in Older Adults without Sun Exposure: A Randomized Double‐Blind Trial. Journal of the American Geriatrics Society. 64(1): 65-72.
- Bingham, C.T. and L.A. Fitzpatrick. (1993). Noninvasive testing in the diagnosis of osteomalacia. The American journal of medicine. 95(5): 519-523.
- Murray, S.E. (2013). Timing of symptom improvement after parathyroidectomy for primary hyperparathyroidism. Surgery. 154(6): 1463-1469.
- Anisiewicz, A. (2018). Unfavorable effect of calcitriol and its low-calcemic analogs on metastasis of 4T1 mouse mammary gland cancer. International journal of oncology. 52(1): 103-126.
- Ghazizadeh, T. (2015). EVALUATION OF ANTIOXIDANT ACTIVITY OF KLOTHO IN HEMODIALYSIS PATIENTS AND RENAL TRANSPLANT RECIPIENTS. URMIA MEDICAL JOURNAL. 26(9): 743-753.
- Petersen, M.A. (2006). Addendum to the EORTC QLQ-C30 Scoring Manual: Scoring of the EORTC QLQ-C15-PAL.
- Fayers, P.M. (1999). EORTC QLQ-C30 scoring manual.
- Groenvold, M. (2006). The development of the EORTC QLQ-C15-PAL: a shortened questionnaire for cancer patients in palliative care. European Journal of Cancer. 42(1): 55-64.
- Scott, N.W. (2010). Differential item functioning (DIF) analyses of health-related quality of life instruments using logistic regression. Health and quality of life outcomes. 8(1): 81.
- Petersen, M.A. (2006). Item response theory was used to shorten EORTC QLQ-C30 scales for use in palliative care. Journal of Clinical Epidemiology. 59(1): 36-44.
- Koo, K. (2013). Quality of life in patients with brain metastases receiving upfront as compared to salvage stereotactic radiosurgery using the EORTC QLQ-C15-PAL and the EORTC QLQ BN20+ 2: a pilot study. Journal of Radiation Oncology. 2(2): 217-224.
- Yellen, S.B. (1997). Measuring fatigue and other anemia-related symptoms with the Functional Assessment of Cancer Therapy (FACT) measurement system. Journal of pain and symptom management. 13(2): 63-74.
- Okada, K.I. (2018). Prospective validation of patient fatigue questionnaire (FACIT‑F) for fatigue assessment in nab‑paclitaxel plus gemcitabine therapy. Molecular and clinical oncology. 8(1): 121-126.
- Webster, K, D. Cella, K. Yost. (2003). The F unctional A ssessment of C hronic I llness T herapy (FACIT) Measurement System: properties, applications, and interpretation. Health and quality of life outcomes. 1(1): 79.
- Harrold, J. (2005). Is the palliative performance scale a useful predictor of mortality in a heterogeneous hospice population? Journal of palliative medicine. 8(3): 503-509.
- Head, B, C.S. Ritchie, T.M. Smoot. (2005). Prognostication in hospice care: can the palliative performance scale help? Journal of palliative medicine. 8(3): 492-502.
- Lau, F. (2006). Use of Palliative Performance Scale in end-of-life prognostication. Journal of palliative medicine. 9(5): 1066-1075.
- Downing, M. (2007). Meta-analysis of Survival Prediction With Palliative Performance Scale/Discussant Response# 1/Discussant Response# 2. Journal of palliative care. 23(4): 245-252.
- Olajide, O. (2007). Validation of the palliative performance scale in the acute tertiary care hospital setting. Journal of palliative medicine. 10(1): 111-117.
- Lau, F. (2008). Use of the Palliative Performance Scale in survival prediction for terminally ill patients in Western Newfoundland, Canada. Journal of palliative care. 24(4): 282-284.
- Mahajan, A.S, P. Agarwal. (2017). Current Perspective on Vitamin D Deficiency: A Mini Review. J Physiol. 30(1): 5-11.
- Abrams, G.D, D. Feldman, M.R. Safran. (2018). Effects of Vitamin D on Skeletal Muscle and Athletic Performance. JAAOS-Journal of the American Academy of Orthopaedic Surgeons. 26(8): 278-285.
- Kvaran, R.B. (2018). Vitamin D Status in Critically Ill Patients.
- Gaikwad, M. (2018). Factors associated with vitamin d testing, deficiency, intake, and supplementation in patients with chronic pain. Journal of dietary supplements. 15(5): 636-648.
- Feldman, D. (2014). The role of vitamin D in reducing cancer risk and progression. Nature reviews cancer. 14(5): 342-357.
- Brakta, S. (2015). Role of vitamin D in uterine fibroid biology. Fertility and sterility. 104(3): 698-706.
- Ahmed, J.H. (2019). Vitamin D Status and Association of VDR Genetic Polymorphism to Risk of Breast Cancer in Ethiopia. Nutrients. 11(2): 289.
- Galunska, B. (2018). VITAMIN D: A POTENTIAL BIOMARKER FOR MORE AGGRESSIVE PROSTATE CANCER. Biological Markers in Fundamental and Clinical Medicine (collection of abstracts). 2(2): 23-24.
- Edwards, B.J.-A. (2018). Vitamin D deficiency in older cancer patients with solid tumors, effect on overall survival. American Society of Clinical Oncology.
- Andersen, M.R. (2019). Effects of Vitamin D Use on Health-Related Quality of Life of Breast Cancer Patients in Early Survivorship. Integrative cancer therapies. 1534735418822056.
- Madden, J. (2018). De novo vitamin D supplement use post-diagnosis is associated with breast cancer survival. Breast cancer research and treatment. 172(1): 179-190.
- Bergman, P. (2015). Vitamin D supplementation improves well-being in patients with frequent respiratory tract infections: a post hoc analysis of a randomized, placebo-controlled trial. BMC research notes. 8(1): 498.
- Sepehrmanesh, Z. (2015). Vitamin D Supplementation Affects the Beck Depression Inventory, Insulin Resistance, and Biomarkers of Oxidative Stress in Patients with Major Depressive Disorder: A Randomized, Controlled Clinical Trial, 2. The Journal of nutrition. 146(2): 243-248.
- Wu, Z. (2016). Effect of vitamin D supplementation on pain: a systematic review and meta-analysis. Pain Physician. 19(7): 415-427.
- Mohr, S.B. (2014). Meta-analysis of vitamin D sufficiency for improving survival of patients with breast cancer. Anticancer research. 34(3): 1163-1166.
Download Provisional PDF Here
PDF




p (1).png)




.png)




.png)
